  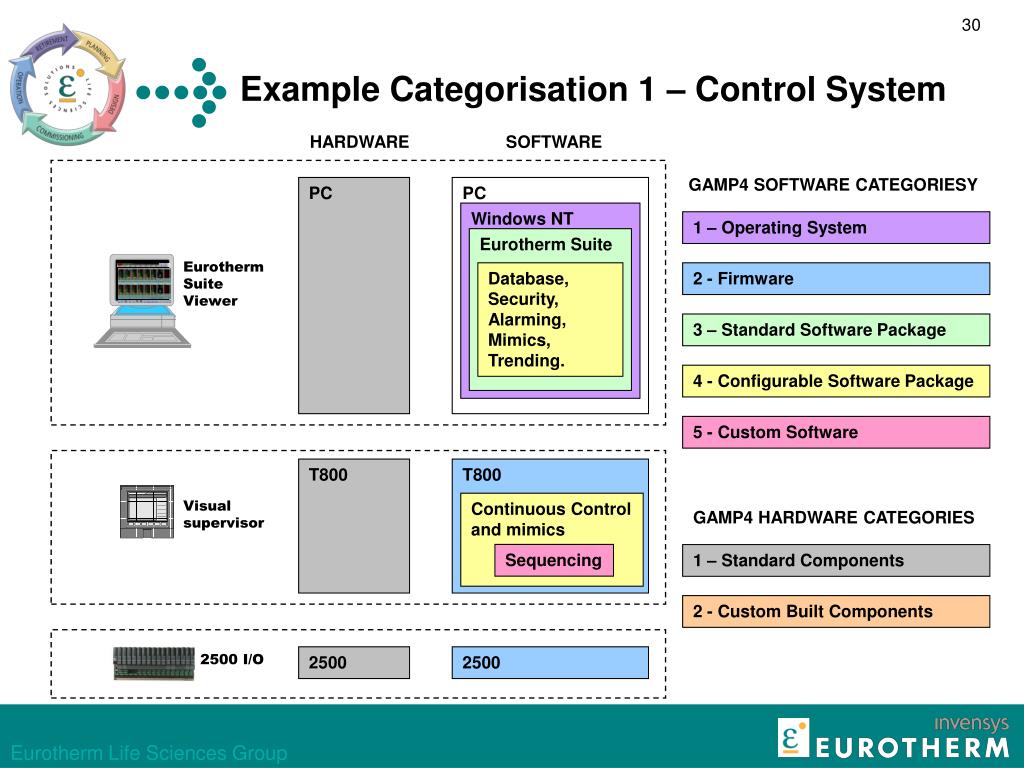 
Under this new standard, the IT and Infrastructure supporting components life cycle should focus on installation verification and assess/record the tool adequacy for use. This change in the software category for IT and Infrastructure supporting components aligns very much with the new FDA CSA draft guidance soon to be released. The above was an expected significant update. “Software, systems, and tools supporting computerized system life cycle activities and IT and infrastructure processes (as opposed to supporting business and pharmaceutical and medical device life cycle processes)” (GAMP5 2 nd Ed, 2022).Per the new edition, software in Category 1 includes: IT and Infrastructure Supporting Software, Systems, and Tools are now Software Category 1 As usual, the validation approach decision and the rationale should be documented. With this new GAMP 5 edition, regulated companies should fully embrace the scale-up or down the life cycle activities following a risk-based approach. This hesitant response could result from two factors: 1) regulated companies choose a play-safe strategy by following a conservative mindset to CSV and generating all validation deliverables according to procedure per system category, or 2) lacking knowledge and resources on how to implement an effective quality risk-management approach. However, through all these years, regulated companies have been timid to embrace it fully. The risk-based approach concept was central to GAMP5 First Edition (2008). “The software category is just one factor in a risk-based approach the life cycle activities should be scaled based on the overall GxP impact, complexity, and novelty of the system (derived from the criticality of the business process supposed by the system)” (GAMP5 2 nd Ed, 2022)įollowing a risk-based approach for computerized systems validation is not a stranger for regulated companies.Life cycle activities scaling based on other factors besides software category GAMP5 2 nd Edition authors made a key point highlighting that “categorization is not intended to provide a checklist approach to validation.” (GAMP5 2 nd Ed, 2022) From this perspective, regulated companies should apply critical thinking to determine the validation approach for the computerized system implementation, rather than use categories (especially software categories) as a cookie cutter prescription to define the validation deliverables required for the system implementation. Therefore, regulated companies should not ‘rigidly stick’ a computerized system into a single category to determine the approach and extent of the validation exercise or the computer system lifecycle. “Computerized systems are generally made up of a combination of components from different categories the categories should be viewed as a continuum” (GAMP5 2 nd Ed, 2022).Software & Hardware Categories Viewed as a Continuum This appendix has significantly changed from GAMP 5 First Edition (2008). This first post contains updates related to Appendix M4 – Categories of Software & Hardware. In this GAMP5 2nd Edition & You quick read series, Xevalics Consulting aims to highlight content updates in this new edition that may impact your current computer system validation (CSV) program.īefore continuing, we would like to recognize the extraordinary effort of leading authors who dedicated their time to having this publication ready in about a year (this is quite an accomplishment!) The ISPE GAMP® 5: A Risk-Based Approach to Compliant GxP Computerized Systems (Second Edition) has been recently published. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
